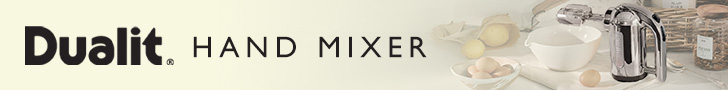ASX listed company, G Medical Innovation has developed a smartphone case that not only protects your phone but monitors your health and wellbeing with users able to send their medical information to third parties such as family members or medical professionals.
The Prizma Medical Smartphone Case is used in conjunction with its app via Bluetooth measures a user’s heart rate, stress level, ECG, body temperature and oxygen saturation.
The case was developed in Israel and can be made for the iPhone 7, Alcatel Idol 4 and Samsung Galaxy S7.
G Medical Innovations says the Prizma case improves productivity and efficiency to improve health outcomes and continuously collects, consolidates and analyses medical data collecting trends overtime.
The Prizma Medical Smartphone Case has been accepted to the Green Channel expedited Guangdong China Food and Drug Administration (CFDA) regulatory approval process through yhe company’s Chinese based subsidiary Guangzhou Yimei Innovatice Medical.
This means the company will be able to move forward quickly towards final regulatory CFDA approval and commercialisation of the smartphone case. The special review and approval procedures put in place for innovative medical devices by the CFDA ensures and verifies the safety and effectiveness of products for the vast Chinese market, the company says this represents a significant milestone for G Medical and its planned rollout in such an important market.
Read more about the smartphone case here. The case is available for pre-order at US$249.99 with an additional US$4.99 a month for a medical history cloud service.
ChannelNews understands the case will be available in Australia in a couple of months.